Abstract
Introduction
Relapses are inevitable in patients with newly diagnosed multiple myeloma (NDMM), necessitating multiple lines of therapy. Achievement of complete response (CR) after first line therapy has been previously shown to predict longer survival. We aimed to study the prognostic impact of achieving CR after first line therapy on survival after relapse among patients with NDMM.
Methods
Data from 2,707 consecutive patients with NDMM seen at Mayo Clinic, within 90 days of diagnosis, between 2004 and 2015, was analyzed retrospectively. Of these, 381 (14.1%) patients were lost to follow-up. The data for response assessment after first line therapy was available for 1,636 patients. Among these, 1,079 patients experienced a relapse mandating the start of second line therapy and form the current study population. The progression free survival (PFS) was calculated from the date of start of first line therapy or second line therapy till progression or death from any cause or the date of last follow up. The overall survival (OS) was calculated from the date of start of second line therapy till death from any cause or the date of last follow up. The survival curves were generated using Kaplan-Meier method and their differences were estimated using log rank test.
Results
Among the 1,079 patients who experienced first relapse, patients who previously achieved CR (N=190; 17.6%) were compared with those who did not achieve CR (n=889; 82.4%) as the best hematological response after first line treatment.
The comparison of the patients with CR and no CR after first line treatment is provided in Table 1, along with baseline characteristics at diagnosis and details of first line treatment. Treatment at relapse consisted of autologous stem cell transplant (ASCT) in 139 patients (12.9%), proteasome inhibitor (PI) based therapy in 153 patients (14.2%), immunomodulator (IMiD) based therapy in 405 patients (37.5%), alkylator based therapy in 34 patients (3.2%), combination of PI and IMiD in 164 patients (15.2%), combination of PI and alkylators in 129 patients (12%), and steroids and other drugs in 22 patients (2%), while 33 patients (3.1%) were either unfit to receive any therapy or refused treatment.
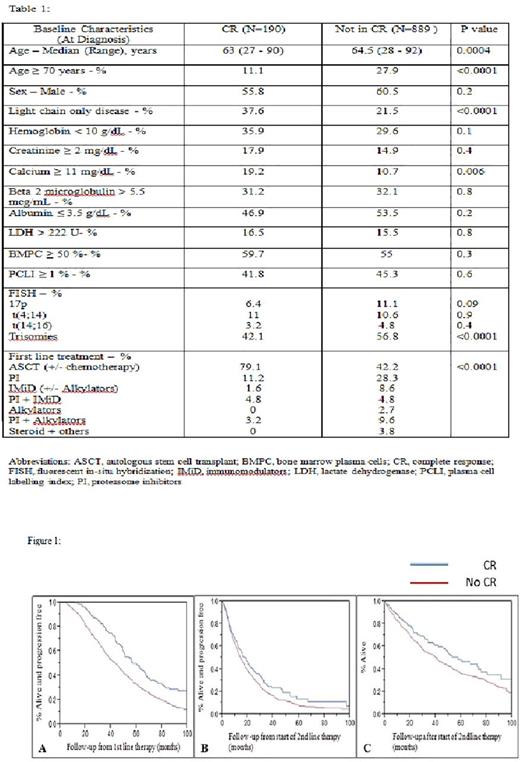
The median follow-up for the entire cohort was 84.1 months (95% CI; 79.3, 89.8) from the start of 1st line treatment and 47.1 months (95% CI; 43.1, 53.1) from the start of 2nd line treatment. During follow-up, 594 patients (55.1%) experienced a second relapse or progression; 97 patients (51.1%) among those who achieved CR and 497 patients (55.9%) among those who did not achieve CR after first line treatment. The median PFS after 1st and 2nd line treatment was significantly longer for patients who achieved CR with first line therapy [60.1 months (95% CI; 51, 67) after 1st line and 17.7 months (95% CI; 13.4, 21.9) after 2nd line] as compared to those who did not achieve CR after first line treatment [43.6 months (95% CI; 40,46.8) after 1st line and 14.5 months (95% CI; 13, 16) after 2nd line] (p<0.0001 for PFS after 1st line and p = 0.04 for PFS after 2nd line; Figure 1A and IB). Similarly, the median OS after 2nd line treatment was also significantly longer for patients who achieved CR [58.1 months (95% CI; 45.5, 75.6)] as compared to those who did not achieve CR [40.5 months (95% CI; 34.9, 45.4)] after 1st line treatment (p = 0.01; Figure 1C).
Conclusion
Patients with NDMM fare well post relapse/progression. Achievement of CR after first line treatment delays progression and predicts improved survival even after relapse, reflecting the contribution of disease biology to outcomes seen in MM. This novel information could be useful in planning better salvage therapies in the challenging patient population with relapsed disease.
Sidana: Janssen: Honoraria. Kapoor: Takeda, Celgene and Amgen: Research Funding. Dispenzieri: Celgene, Millenium, Pfizer, Janssen: Research Funding. Gertz: Millennium: Consultancy, Honoraria; Celgene, Novartis, Smith-Kline, Prothena, Ionis, Amgen: Honoraria. Dingli: Janssen: Consultancy; Millenium: Consultancy; Karyopharm Therapeutics: Research Funding; Takeda: Consultancy; Alexion Pharmaceuticals: Consultancy. Russell: Imanis Life Sciences: Equity Ownership; Vyriad: Equity Ownership. Kumar: Celgene, Millennium, BMS, Onyx, Janssen, Noxxon, AbbVie, Amgen, Merck, Oncopeptides, Skyline Diagnostics, Takeda: Consultancy; Celgene, Millennium/Takeda, Onyx, AbbVie, Janssen, Sanofi, Novartis, Amgen, Genentech, Merck, Oncopeptides, Roche, Skyline Diagnostics: Research Funding; Skyline: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal